the Energy (Im)Balance of Our Current Atmosphere.
|
Introduction
To Global Change I Lecture Notes |
|
|||
| Natural Climate Change | Radiative Forcing | Global Warming Potential | Summary | |
Driving Questions:
- What has controlled past changes in Earth's temperature?
- What processes lead to temperature change?
- What gases comprise the atmosphere and how do they influence the temperature?
- What are the global warming potentials for greenhouse gases?
1. Natural Climate Change
We believe that the temperature of the earth has varied wildly over the evolution of the earth. Figure 1 shows an estimate of temperature changes as complied by Scotese. So how can it be that the climate has changed so over the ages and what processes could lead to these changes?
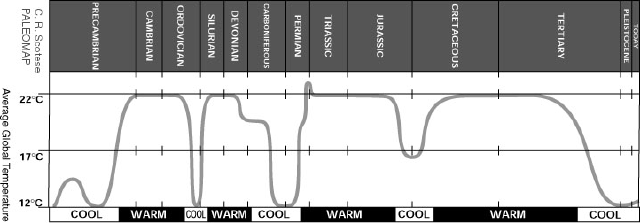
Figure 1. Estimated changes in global temperature.
The processes for changing climate naturally over long timescales include:
-
Plate tectonics
-
Long-term carbon cycle
-
Solar Variations, and
-
Orbital Variations
Plate Tectonics
The movement of the continents has obviously influenced the climate at specific locations (Figure 2), but could also influence the global temperature by redistributing the collection of solar radiation and/or providing land masses on which continental glaciers could form.

Figure 2. Location of continents during the Devonian
period from Scotese.
Long-term carbon cycle
On long timescales of millions of years, the CO2 composition of Earth's atmosphere has been controlled by the exchange of carbon between the atmosphere, life, and rocks. Volanic eruptions (both on land and in the ocean) and metamorphism are sources of CO2 to the atmosphere. Silicate weathering and organic carbon burial are processes that have consumed atmospheric CO2. The balance between these three processes have maintained Earth's climate within a habitable zone.
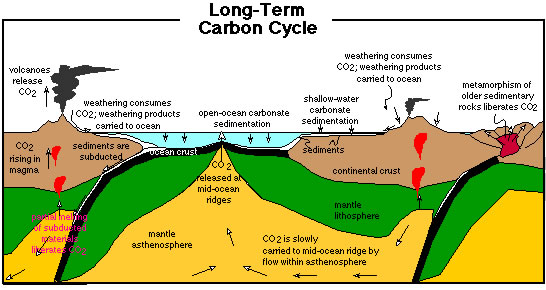
Figure 3. The long-term (inorganic) carbon cycle (Bice, 2001). Missing from this figure is the portrayal of organic carbon burial.
Solar Variability
On geological timescales of millions of years, the Sun's luminosity has increased at about 1% per 100 million years. It is surprising then that Earth's climate has remained relatively stable. It is thought that the increase in solar radiation has been balanced by a long-term decrease in atmosphere CO2 through an increase in silicate weathering.
Orbital Variability
The Earth's orbit changes over time in ways that could influence the amount of energy received at the surface. Thes e include changes in eccentricity, precession of the equinox, and changes in the Earth's tilt (obliquity).
Eccentricity
|
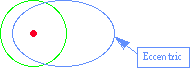 |
Precession of the Equinox
|
 |
Obliquity
|
2. Radiative Forcing
The temperature of the Earth's surface and atmosphere are dictated by
a balance between incoming energy and outgoing energy. When more energy
is received than lost, temperatures rise. The Earth's surface, for example,
absorbs radiation from the Sun. This energy is then redistributed by the
atmospheric and oceanic circulations and radiated back to space at longer
(infrared) wavelengths. For the annual mean and for the Earth as a whole,
the incoming solar radiation energy is balanced approximately by the outgoing
terrestrial radiation. Any factor that alters the radiation received from
the Sun or lost to space, or that alters the redistribution of energy
within the atmosphere and between the atmosphere, land, and ocean, can
affect climate. A change in the net radiative energy available to the
global Earth-atmosphere system is termed a radiative forcing. Positive
radiative forcings tend to warm the Earth’s surface and lower atmosphere.
Negative radiative forcings tend to cool them.
Increases in the concentrations of greenhouse gases will reduce the efficiency
with which the Earth’s surface radiates to space. More of the outgoing
terrestrial radiation from the surface is absorbed by the atmosphere and
re-emitted at higher altitudes and lower temperatures. This results in
a positive radiative forcing that tends to warm the lower atmosphere and
surface. Because less heat escapes to space, this is the enhanced greenhouse
effect – an enhancement of an effect that has operated in the
Earth’s atmosphere for billions of years due to the presence of naturally
occurring greenhouse gases: water vapor, carbon dioxide, ozone, methane
and nitrous oxide. The amount of radiative forcing depends on the size
of the increase in concentration of each greenhouse gas, the radiative
properties of the gases involved (indicated by their global warming
potential), and the concentrations of other greenhouse gases already
present in the atmosphere. Further, many greenhouse gases reside in the
atmosphere for centuries after being emitted, thereby introducing a long-term
commitment to positive radiative forcing.
Anthropogenic aerosols (microscopic airborne particles or droplets) in
the troposphere, such as those derived from fossil fuel and biomass burning,
can reflect solar radiation, which leads to a cooling tendency in the
climate system. Because it can absorb solar radiation, black carbon (soot)
aerosol tends to warm the climate system. In addition, changes in aerosol
concentrations can alter cloud amount and cloud reflectivity through their
effect on cloud
properties and lifetimes. In most cases, tropospheric aerosols tend to
produce a negative radiative forcing and a cooler climate. They have a
much shorter lifetime (days to weeks) than most greenhouse gases (decades
to centuries), and, as a result, their concentrations respond much more
quickly to changes in emissions. Volcanic activity can inject large amounts
of sulphur-containing gases (primarily sulfur dioxide) into the stratosphere,
which are transformed into sulfate aerosols. Individual eruptions can
produce a large, but transitory, negative radiative forcing, tending to
cool the Earth’s surface and lower atmosphere over periods of a few
years.
When radiative forcing changes, the climate system responds on various
time-scales. The longest of these are due to the large heat capacity of
the deep ocean and dynamic adjustment of the ice sheets. This means that
the transient response to a change (either positive or negative) may last
for thousands of years. Any changes in the radiative balance of the Earth,
including those due to an increase in greenhouse gases or in aerosols,
will alter the global hydrological cycle and atmospheric and oceanic circulation,
thereby affecting weather patterns and regional temperatures and precipitation.
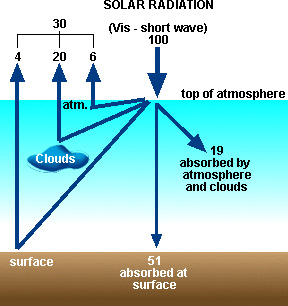 |
| Figure 5. Global average flow of shortwave (solar) radiation through the Earth's atmosphere. |
Shortwave Radiation Budget
Solar radiation entering the Earth's atmosphere (called "shortwave" radiation) can be reflected off clouds, the surface, and air molecules and dust. On a global average this accounts for about 30% of incoming radiation (see Figure 5). This percentage is quantified as the albedo of the system.
Albedo = percentage of incoming radiation that is reflected back into space = 30% for Earth
Another 19% on average is absorbed by the atmosphere, mainly by ozone
in the Earth's stratosphere. The remaining 51% is absorbed by the Earth's
surface.
Over a long term average, the Earth and its atmosphere must radiate as much energy out to space as it receives from the sun, but over the course of a year or a day or as one moves geographically it is likely that such a balance will not be present. At night and in the winter, for example, there is less solar radiation producing an energy deficit and leading to lower temperatures at those times in general.
In order to understand the whole energy balance we must also consider the other means for exchanging energy between the Earth's surface, atmosphere and space.
 |
|||
| Figure 6: Energy exchange between the Earth's
surface and its atmosphere. |
|||
Gains |
Losses |
||
|---|---|---|---|
|
|
|||
| 51 | Visible from Sun | 7 | Conduction, Convection |
| 96 | IR from atmosphere | 23 | Evaporation |
| 117 | IR radiation | ||
|
|
|||
| 147 | net | 147 | net |
Longwave Radiation Budget
As was learned earlier all objects emit radiation in an amount and at a wavelength dictated by the object's temperature. The 51% of shortwave radiation absorbed by the Earth's surface (Figure 5) heats the surface. But as the surface heats it emits radiation in the infrared back into the atmosphere.
Figure 6 shows the annual global average exchange of energy between the
Earth's surface and the atmosphere. Note the 51% of original solar radiation
is absorbed, but 117% of the original solar input is emitted to the atmosphere,
how can this be?
The answer makes sense when we consider that the surface of a planet receives a great deal of energy from its own atmosphere. Thus the effect of the atmosphere is to warm the surface over the temperature above that resulting from the Sun's energy.
The atmosphere warms the Earth by "trapping" radiation, allowing the surface to warm to 300°K. At that temperature, the black body surface radiation is large enough to ensure that an equilibrium condition pertains. The atmosphere traps radiation through the action of certain gases, called Greenhouse Gases. These gases (e.g., CO2, H2O, NO, CFCs, CO) are very good at absorbing and re-emitting infrared radiation. They intercept the IR radiation from the ground and reflect some of the energy back to the ground, warming it up more than would occur otherwise.
3. Global Warming Potential
The Global Warming Potential (GWP) of a greenhouse gas is the ratio of global warming, or radiative forcing – both direct and indirect – from one unit mass of a greenhouse gas to that of one unit mass of carbon dioxide over a period of time. Hence this is a measure of the potential for global warming per unit mass relative to carbon dioxide.
Global Warming Potentials are presented in Table 1 for an expanded set of gases. GWPs are a measure of the relative radiative effect of a given substance compared to CO2, integrated over a chosen time horizon. New categories of gases in Table 1 include fluorinated organic molecules, many of which are ethers that are proposed as halocarbon substitutes. Some of the GWPs have larger uncertainties than that of others, particularly for those gases where detailed laboratory data on lifetimes are not yet available. The direct GWPs have been calculated relative to CO2 using an improved calculation of the CO2 radiative forcing, the SAR response function for a CO2 pulse, and new values for the radiative forcing and lifetimes for a number of halocarbons. Indirect GWPs, resulting from indirect radiative forcing effects, are also estimated for some new gases, including carbon monoxide. The direct GWPs for those species whose lifetimes are well characterized are estimated to be accurate within ±35%, but the indirect GWPs are less certain.
| Table 1. Direct Global Warming Potentials (GWPs) relative to carbon dioxide (for gases for which the lifetimes have been adequately characterized). GWPs are an index for estimating relative global warming contribution due to atmospheric emission of a kg of a particular greenhouse gas compared to emission of a kg of carbon dioxide. GWPs calculated for different time horizons show the effects of atmospheric lifetimes of the different gases. | |||||
| Lifetime | Global Warming Potential | ||||
| (years) | (Time Horizon in Years) | ||||
| GAS | 20 yrs | 100 yrs | 500 yrs | ||
| Carbon Dioxide | CO2 |
|
1
|
1
|
1
|
| Methane | CH4 |
12.0
|
62
|
23
|
7
|
| Nitrous Oxide | N2O |
114
|
275
|
296
|
156
|
| Chlorofluorocarbons |
|
|
|
|
|
| CFC-11 |
55
|
4500
|
3400
|
1400
|
|
| CFC-12 |
116
|
7100
|
7100
|
4100
|
|
| CFC-115 |
550
|
5500
|
7000
|
8500
|
|
| Hydrofluorocarbons |
|
|
|
|
|
| HFC-23 | CHF3 |
260
|
9400
|
12000
|
10000
|
| HFC-32 | CH2F2 |
5
|
1800
|
550
|
170
|
| HFC-41 | CH3F |
2.6
|
330
|
97
|
30
|
| HFC-125 | CHF2CF3 |
29
|
5900
|
3400
|
1100
|
| HFC-134 | CHF2CHF2 |
9.6
|
3200
|
1100
|
330
|
| HFC-134a | CH2FCF3 |
13.8
|
3300
|
1300
|
400
|
| HFC-143 | CHF2CH2F |
3.4
|
1100
|
330
|
100
|
| HFC-143a | CF3CH3 |
52
|
5500
|
4300
|
1600
|
| HFC-152 | CH2FCH2F |
0.5
|
140
|
43
|
13
|
| HFC-152a | CH3CHF2 |
1.4
|
410
|
120
|
37
|
| HFC-161 | CH3CH2F |
0.3
|
40
|
12
|
4
|
| HFC-227ea | CF3CHFCF3 |
33
|
5600
|
3500
|
1100
|
| HFC-236cb | CH2FCF2CF3 |
13.2
|
3300
|
1300
|
390
|
| HFC-236ea | CHF2CHFCF3 |
10
|
3600
|
1200
|
390
|
| HFC-236fa | CF3CH2CF3 |
220
|
7500
|
9400
|
7100
|
| HFC-245ca | CH2FCF2CHF2 |
5.9
|
2100
|
640
|
200
|
| HFC-245fa | CHF2CH2CF3 |
7.2
|
3000
|
950
|
300
|
| HFC-365mfc | CF3CH2CF2CH3 |
9.9
|
2600
|
890
|
280
|
| HFC-43-10mee | CF3CHFCHFCF2CF3 |
15
|
3700
|
1500
|
470
|
| Fully fluorinated species |
|
|
|
|
|
| SF6 |
3200
|
15100
|
22200
|
32400
|
|
| CF4 |
50000
|
3900
|
5700
|
8900
|
|
| C2F6 |
10000
|
8000
|
11900
|
18000
|
|
| C3F8 |
2600
|
5900
|
8600
|
12400
|
|
| C4F10 |
2600
|
5900
|
8600
|
12400
|
|
| c-C4F8 |
3200
|
6800
|
10000
|
14500
|
|
| C5F12 |
4100
|
6000
|
8900
|
13200
|
|
| C6F14 |
3200
|
6100
|
9000
|
13200
|
|
| Ethers and Halogenated Ethers |
|
|
|
|
|
| CH3OCH3 |
0.015
|
1
|
1
|
<<1
|
|
| HFE-125 | CF3OCHF2 |
150
|
12900
|
14900
|
9200
|
| HFE-134 | CHF2OCHF2 |
26.2
|
10500
|
6100
|
2000
|
| HFE-143a | CH3OCF3 |
4.4
|
2500
|
750
|
230
|
| HCFE-235da2 | CF3CHClOCHF2 |
2.6
|
1100
|
340
|
110
|
| HFE-245fa2 | CF3CH2OCHF2 |
4.4
|
1900
|
570
|
180
|
| HFE-254cb2 | CHF2CF2OCH3 |
0.22
|
99
|
30
|
9
|
| HFE-7100 | C4F9OCH3 |
5
|
1300
|
390
|
120
|
| HFE-7200 | C4F9OC2H5 |
0.77
|
190
|
55
|
17
|
| H-Galden 1040x | CHF2OCF2OC2F4OCHF2 |
6.3
|
5900
|
1800
|
560
|
| HG-10 | CHF2OCF2OCHF2 |
12.1
|
7500
|
2700
|
850
|
| HG-01 | CHF2OCF2CF2OCHF2 |
6.2
|
4700
|
1500
|
450
|
Summary
- Greenhouse gases selective absorb infrared radiation, thus trapping energy in the atmosphere.
- The atmosphere radiates energy to the surface at an average rate greater than the rate of incoming solar radiation.
- Each greenhouse gas is characterized by its atmospheric lifetime and global warming potential.
All materials ©
the Regents of the University of Michigan unless noted otherwise.